5 Key Advantages of eRegulatory
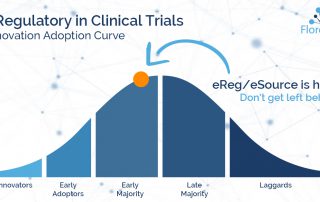
Florence Team2024-12-02T12:50:15-05:00Over 75% of clinical trial sites who have not yet adopted eRegulatory plan to do so in 2019. Why? In addition to growing industry pressure from sponsors who expect a digital site infrastructure, trial sites with eRegulatory platforms are introducing key advantages over those sites without eReg. What are those advantages? Download Five Key Benefits [...]