Florence’s eConsent
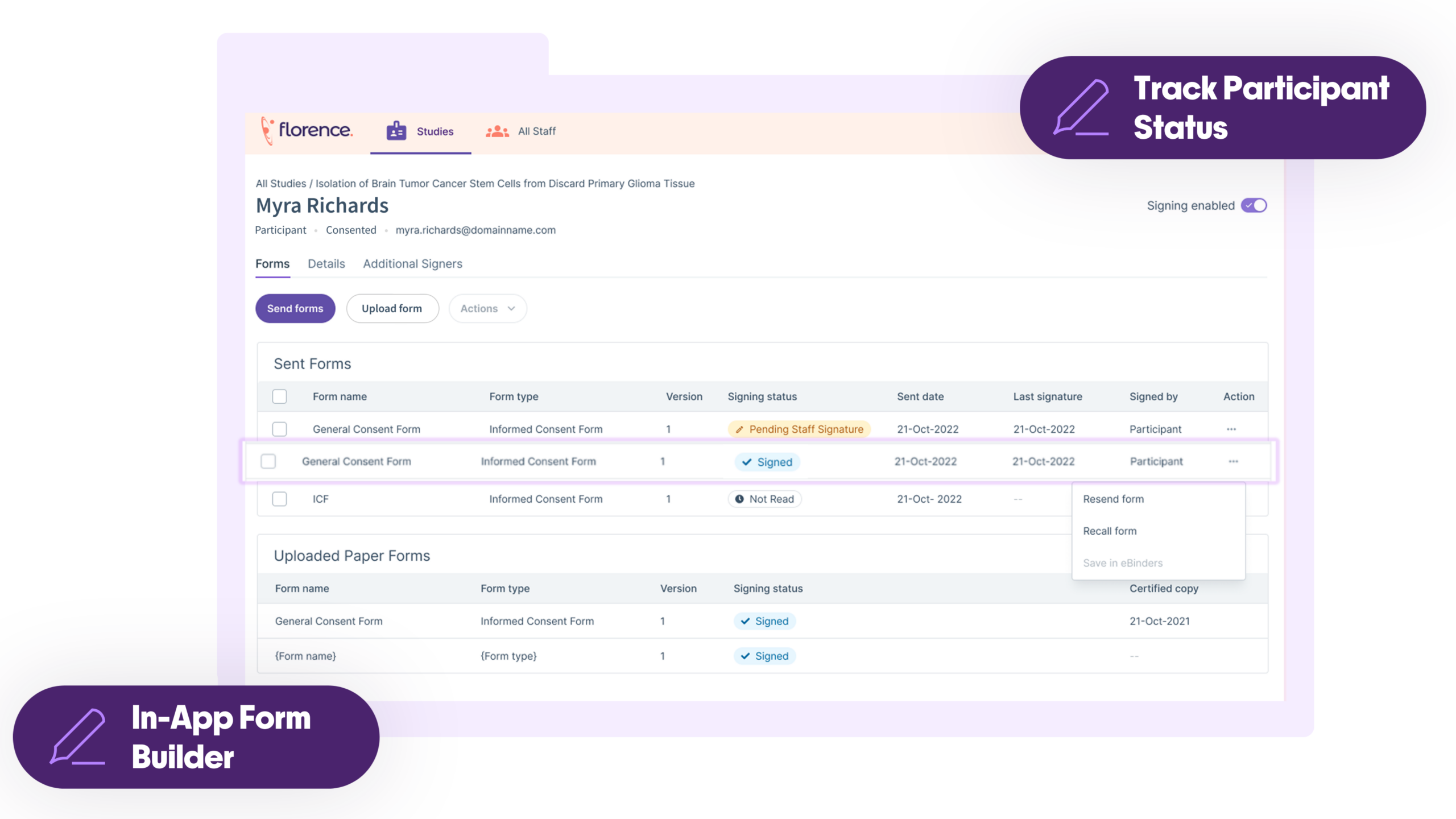
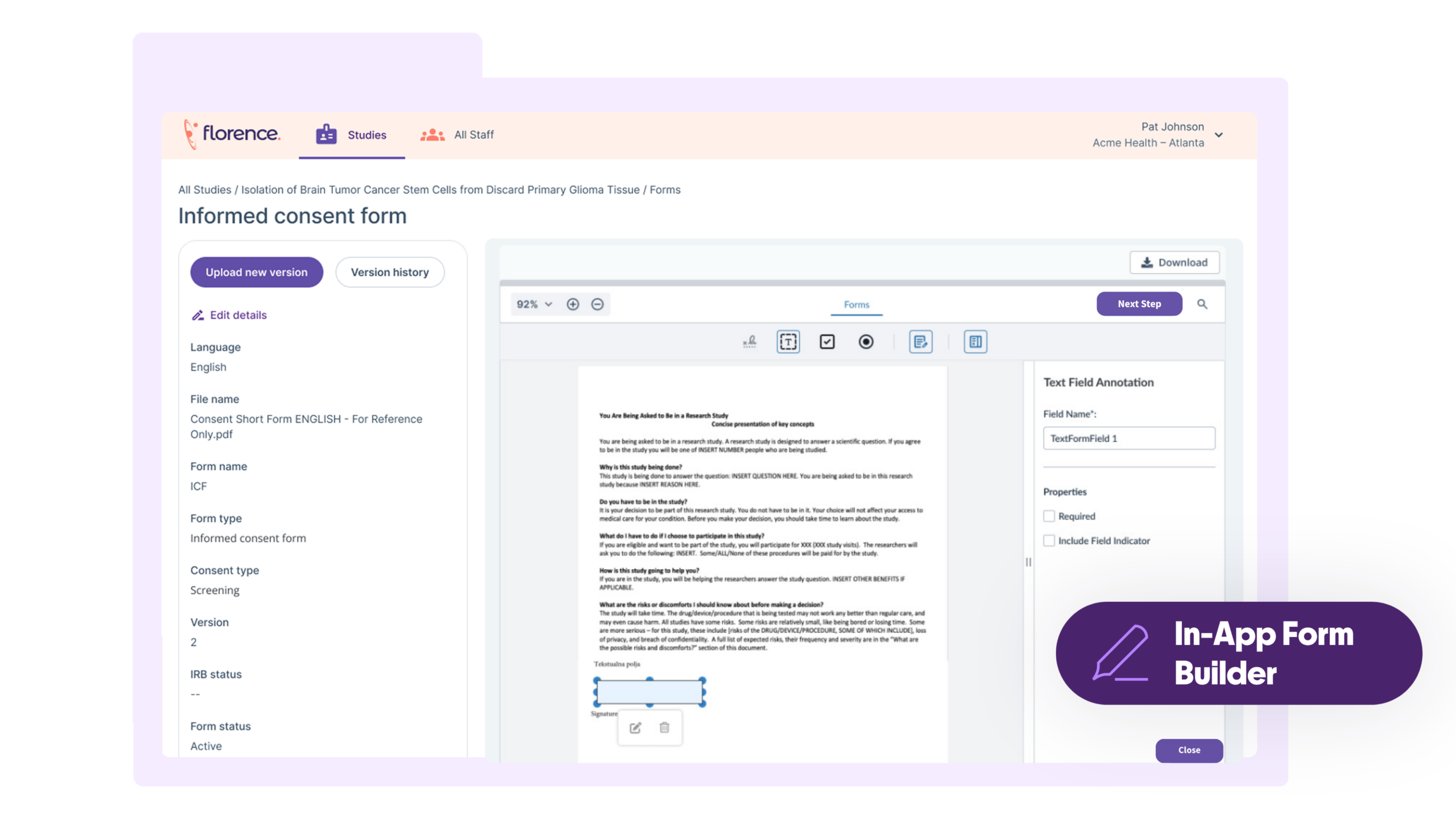
Stop Chasing Paper Consent Forms. Florence eConsent eliminates consenting chaos by embedding digital workflows directly into the platform that 65,000+ sites already use. Get flexible consent options, real-time visibility, and audit-ready data integrity without burdening sites with another disconnected system.
See how it works
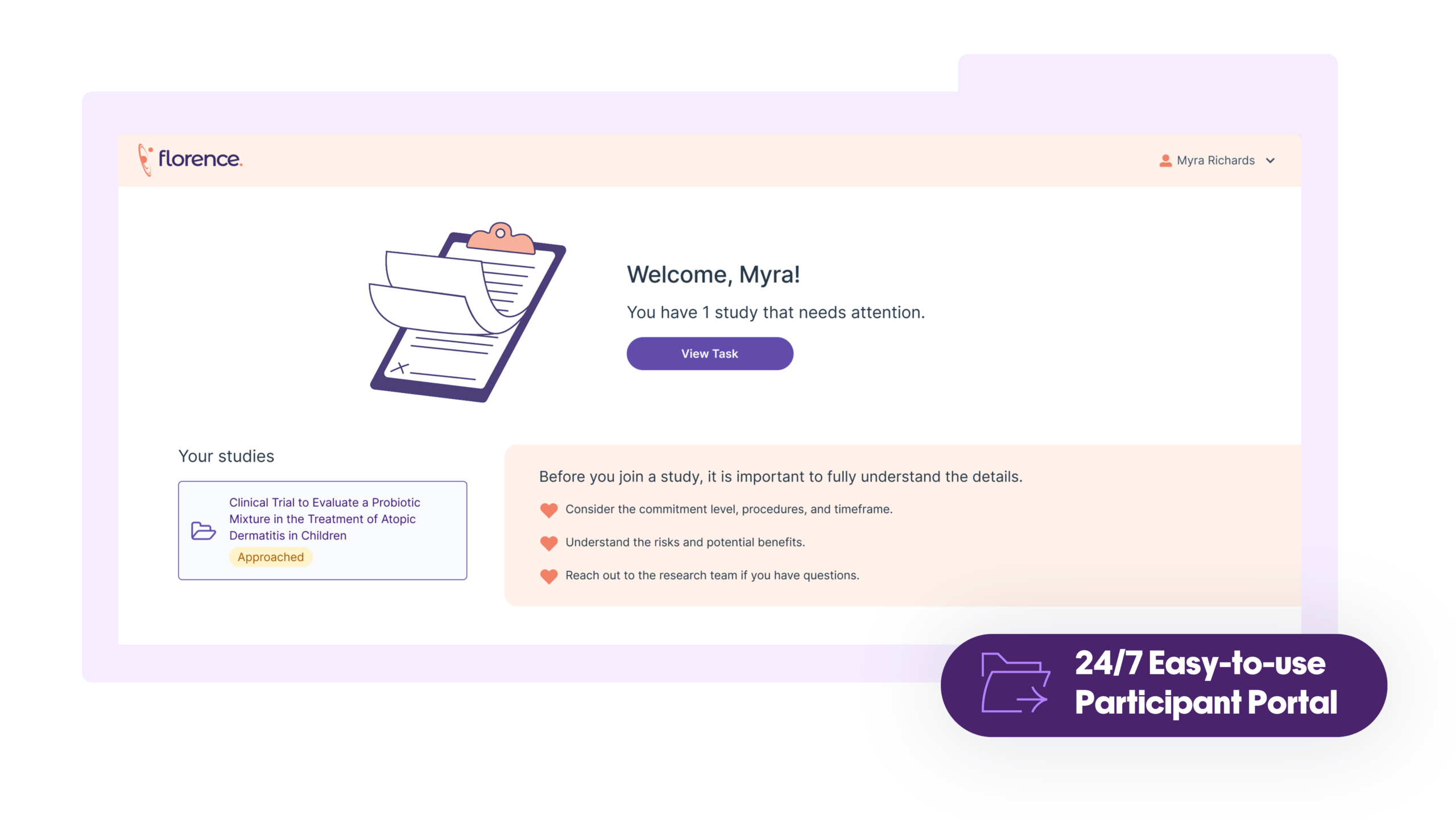
Flexible Workflows for Faster Enrollment
Expand your enrollment population without opening new sites or adding new systems and keep patients engaged
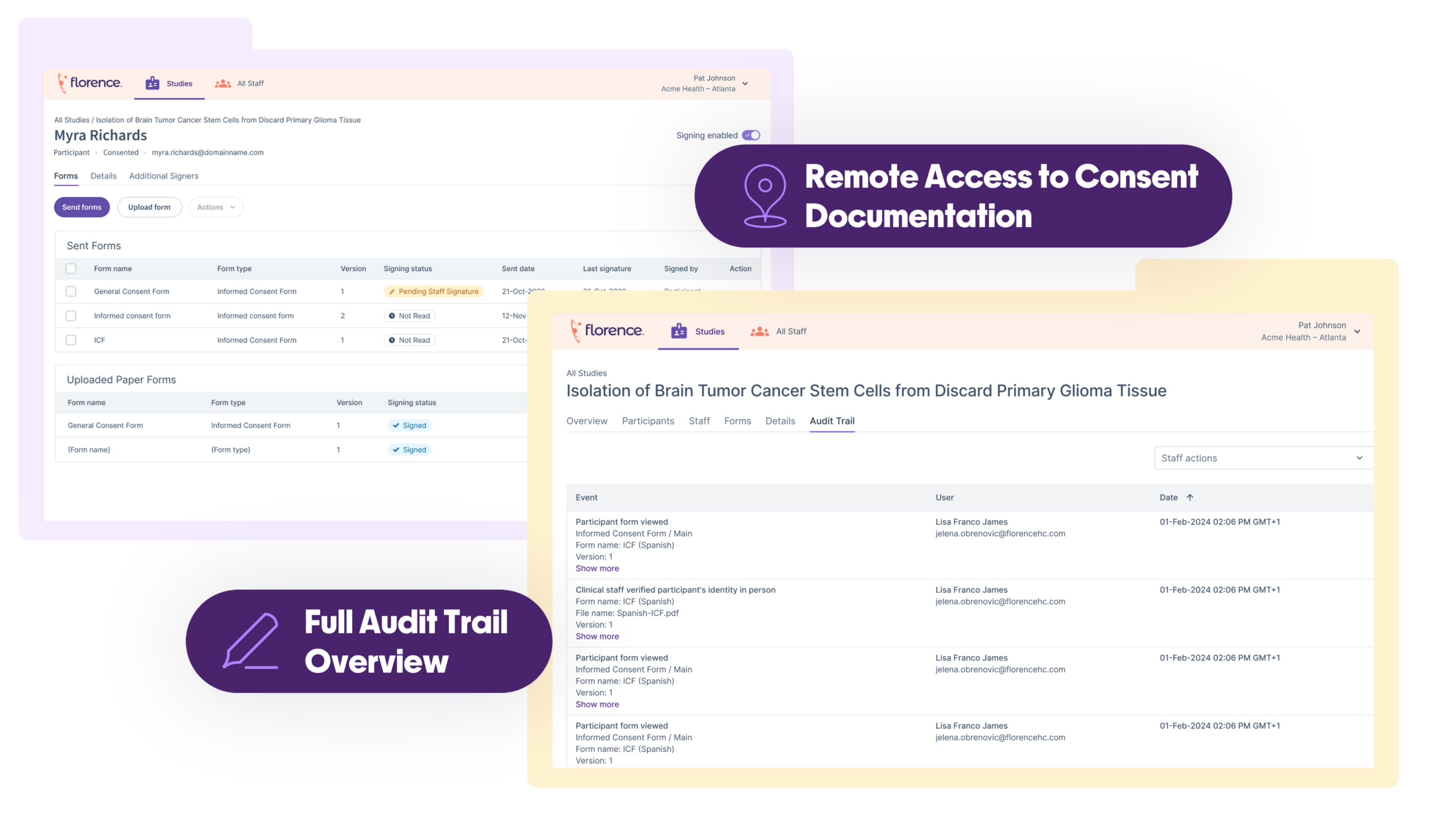
Remote Monitoring to Cut Costs
Eliminate unnecessary site visits and reduce monitoring overhead without sacrificing oversight
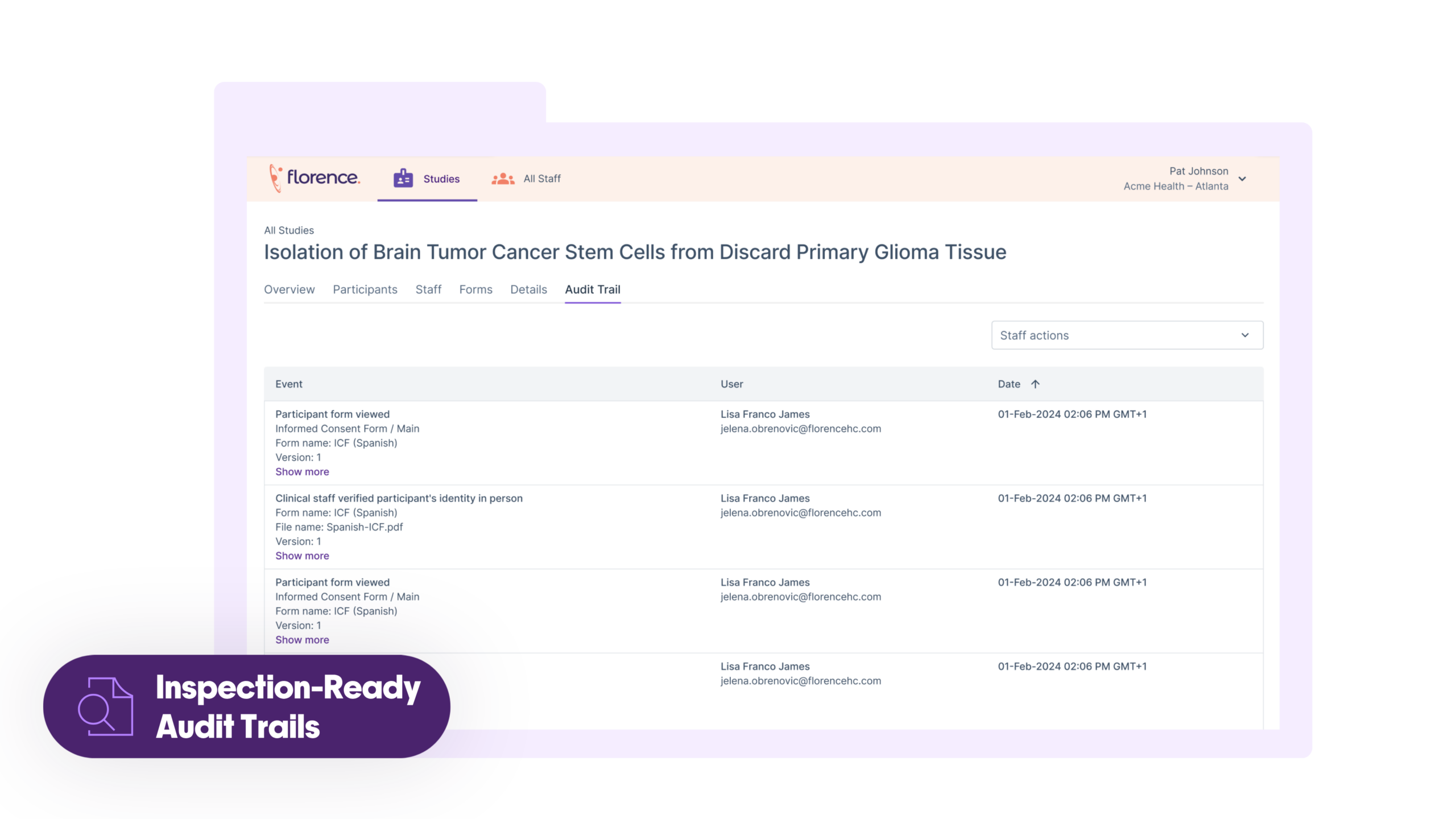
Compliance Built-In to Lower Risk
Reduce regulatory exposure with automated version control and inspection-ready audit trails
Integrate with the tools you use every day.
In clinical research, compliance is crucial. We’ve got you covered globally.